Functional Genomics of Cancer Immunity
Background
The immune system plays a pivotal role in protecting the body from cancer by identifying and eliminating tumor cells. However, as tumors evolve, they accumulate mutations and chromosomal alterations that reshape their ability to trigger immune responses—a phenomenon known as tumor immunogenicity. While some tumors exploit these genomic changes to evade immune detection, others become more immunogenic, increasing their susceptibility to immune surveillance and immunotherapy.
A central question driving our research is: How do DNA repair mechanisms and genomic instability influence the dynamic interplay between cancer cells and the immune system?
To address this, we integrate high-throughput functional screens—both in vitro and in vivo—with advanced molecular and cellular biology approaches. Our goal is to identify DNA damage response (DDR) factors and single nucleotide variants (SNVs) that impact cancer immunity and response to immunotherapy. Ultimately, we aim to translate these discoveries into innovative immuno-oncology strategies that enhance immune recognition and overcome tumor resistance to therapy.
Ongoing Research Projects
1. Mapping the regulators of tumor immunogenicity
Tumors with DNA repair defects can trigger immune responses via cytosolic DNA, increased mutational burden, and immunogenic cell death. However, DDR factors regulating these processes remain largely uncharacterized. To identify such factors, we use CRISPR-Cas9 screening platforms developed in our lab, which systematically quantify the impact of DDR factors and their genetic interactions on immune signaling. By identifying key regulators of tumor immunogenicity, we aim to discover novel therapeutic targets to enhance immune recognition of tumors.
2. Deciphering the impact of SNVs on cancer immunity
Many cancer-associated SNVs remain poorly understood and are classified as variants of uncertain significance. Determining their impact on immunotherapy response is crucial for identifying new biomarkers and therapeutic opportunities. Using CRISPR-dependent base editing, we introduce cancer-associated SNVs into DDR and immune-related genes at scale, assessing their impact on DNA repair, immune signaling, and tumor immunogenicity. This approach aims to pinpoint mutations driving immune evasion and therapy resistance, enabling more effective and personalized cancer treatments.
3. Defining the role of DDR in primary human CD8+ T cells
Therapy-induced DNA damage can impair CD8+ T cell activity, which is crucial for anti-tumor immunity. Understanding how these cells respond to genotoxic stress during cancer treatment is crucial for developing effective combinatorial strategies that integrate DNA-damaging therapies with immunotherapy. We are particularly interested in identifying the DNA repair pathways that sustain T cell functionality under these conditions, with the goal of enhancing T cell resilience and improving immunotherapy outcomes.
Photogallery
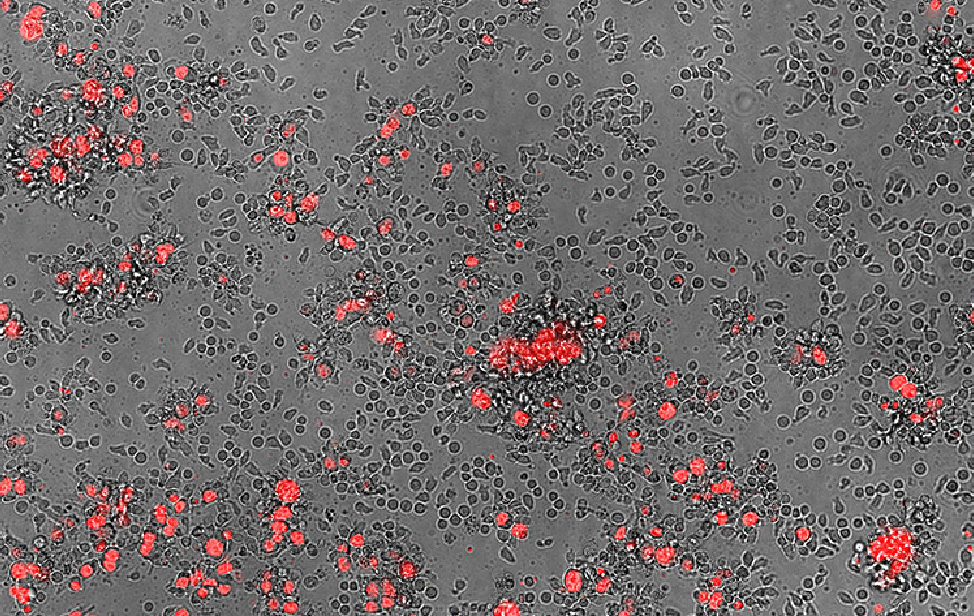 A composite fluorescence and phase contrast microscopy image showing human CD8+ T cells and MDA-MB-436 breast cancer cells (red). The red fluorescence highlights the cancer cells, while the phase contrast reveals the T cells and their interactions with the cancer cells.
A composite fluorescence and phase contrast microscopy image showing human CD8+ T cells and MDA-MB-436 breast cancer cells (red). The red fluorescence highlights the cancer cells, while the phase contrast reveals the T cells and their interactions with the cancer cells. Alkaline comet assay revealing DNA breaks in MDA-MB-436 breast cancer cells after treatment with the DNA-damaging agent Camptothecin (CPT). The comet-like tails indicate the extent of DNA damage.
Alkaline comet assay revealing DNA breaks in MDA-MB-436 breast cancer cells after treatment with the DNA-damaging agent Camptothecin (CPT). The comet-like tails indicate the extent of DNA damage.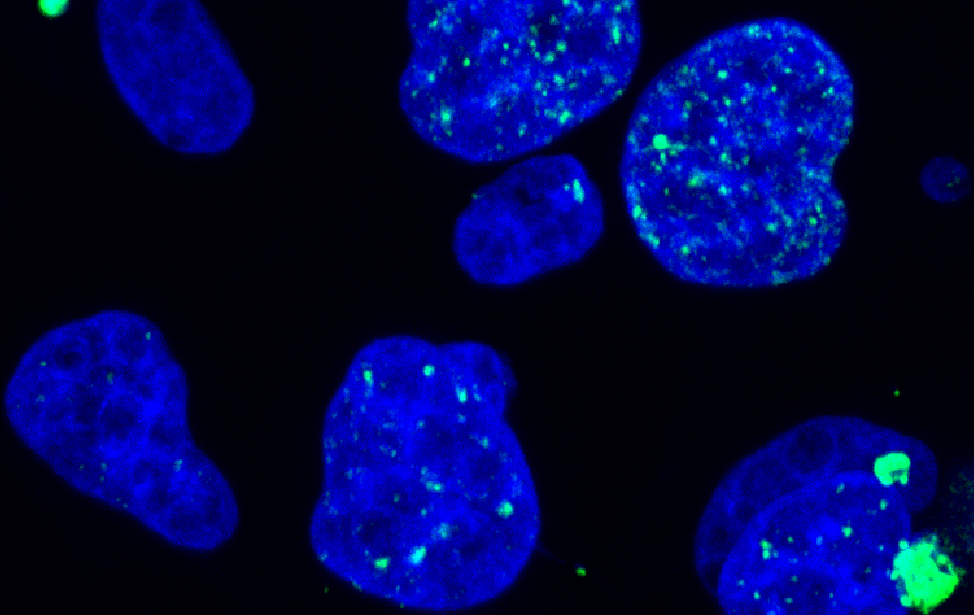 Immunofluorescence staining of MDA-MB-436 breast cancer cells showing γH2AX foci (green), marking DNA damage response activation. Nuclei are counterstained with DAPI (blue), revealing their shape and micronuclei.
Immunofluorescence staining of MDA-MB-436 breast cancer cells showing γH2AX foci (green), marking DNA damage response activation. Nuclei are counterstained with DAPI (blue), revealing their shape and micronuclei.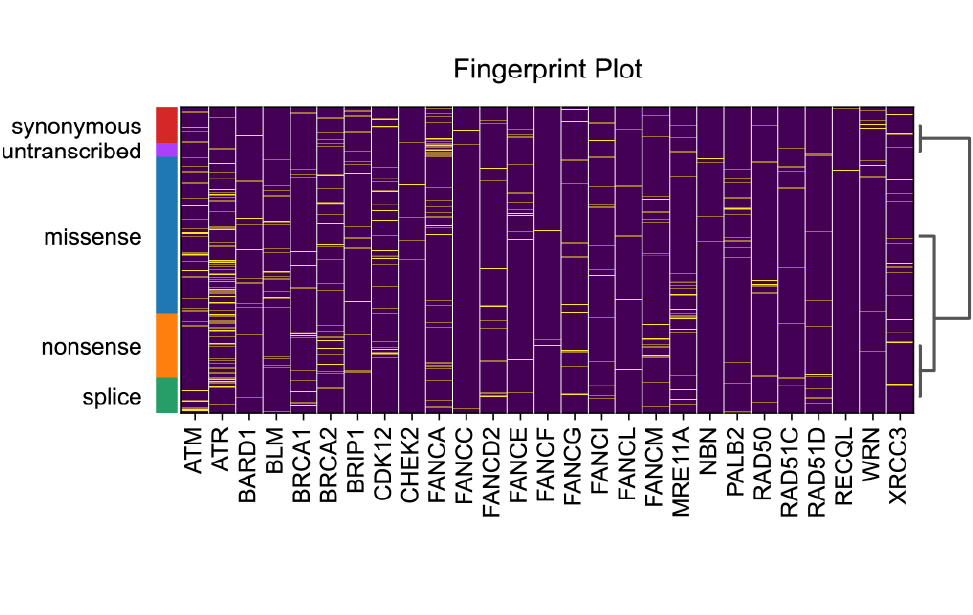 Fingerprint plot of mutation data resulting from CRISPR-mediated base editing screens in DDR (DNA damage response) genes.
Fingerprint plot of mutation data resulting from CRISPR-mediated base editing screens in DDR (DNA damage response) genes.